
OR
Covid-19 vaccination in Nepal
Rethinking Covishield vaccine efficacy
Published On: February 13, 2021 11:30 AM NPT By: Dr Mahesh K Maskey

Dr Mahesh K Maskey
The author, former Ambassador of Nepal to China, is also a former Vice President of International Physicians for Prevention of Nuclear Warnews@myrepublica.com
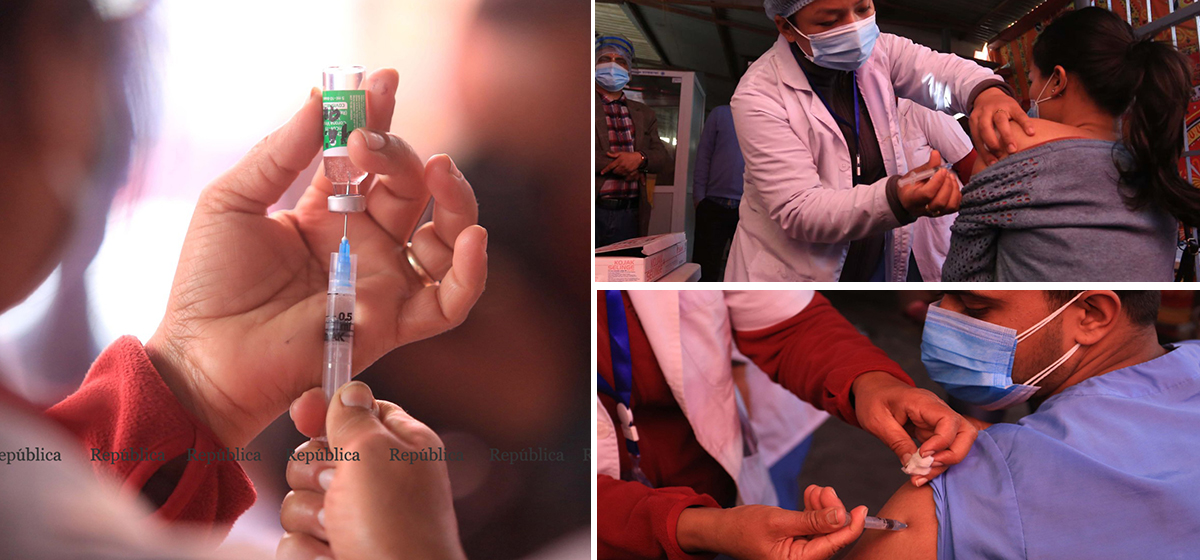
Nepal has completed first phase of Covishield vaccination successfully and has paused for the second dose to be given after 28 days. Nepal should go for single dose and longer interval.
When Covishield (AZD1222) vaccine was rolled out in Nepal, India, the United Kingdom and other parts of the world, there were concerns about its efficacy related to dose schedule. The UK, the first to start this vaccination, opted for longer second dose interval of 12 weeks revising its initial announcement of 28 days. India and Nepal, even though their vaccination program began after the UK, had stuck to 28 days interval schedule.
The recent findings from primary analysis of a larger data set of ongoing third phase of vaccine clinical trial have brought clarity and good news for the UK. Amidst strong criticism from the scientific community, UK’s Scientific Advisory Group for Emergencies (SAGE) had reluctantly endorsed the decision of the government confessing that it was a “very difficult and finely balanced decision”. Such hesitant decision and the opposition from a substantial number of scientists may have influenced India’s and Nepal’s decision to carry on with shorter interval schedule.
Now such criticism has toned down in the light of new evidences which are in favor of UK government decision. On hindsight, one can speculate that the decision of SAGE must have been based upon an Oxford-AstraZeneca confidential report, similar to what is now made public in the The Lancet. These evidences are also relevant to Nepal and its cohort countries where this vaccination program has been launched, and call for a revision of dose interval schedule .In Nepal, the second dose has not been given yet, and a time window is still open for the vaccine advisory committee of Ministry of Health and Population to make the right decision.
True, evidences from vaccine efficacy research are often not clear—either not clearly presented or not clearly understood. The scenario is akin to confusing and conflicting information generated by the researches and modeling on SARS COV-2 virus infection. New evidences that are emerging in the light of more comprehensive analysis of additional data are apparently paradoxical to the commonsense. But with a little patience the underlying logic will make itself visible.
The vaccine had received “emergency” approval based on the interim analysis results obtained from 133 symptomatic cases and about 17000 seronegative participants from an ongoing multi-country third phase clinical trial. Primary analysis of 332 cases on the same participants was published in the preprint of The Lancet journal a few days ago. The countries involved are still UK and Brazil. The data of India are not included here.
The findings suggest that a single dose is much more efficacious on an average (76 percent) for a period of 4 to 12 weeks than the two full dose regimen (62 percent), if the second dose is given before 12 weeks. But if it is given after 12 weeks then the efficacy will rise up to 82 percent. The breakdown of interval is like this: If second dose is given between 4 to 6 weeks the efficacy is only 55 percent, between 6 to 8 weeks (60 percent) and 9-11 weeks (64 percent).
It presents a dilemma. How could a booster second dose actually lower the efficacy in shorter interval than a single dose? And efficacy increase when interval is longer?
Another finding that puzzled the Oxford AstraZeneca vaccine researchers was the enhanced efficacy (90 percent) of an accidental first half dose followed by a second full dose regimen. Though the sample size was small, and it was not given permission for public use by the vaccine regulation committee, it did raise a huge scientific interest and the researchers are planning to do a larger clinical trial, to find answer to this ‘out of the ordinary” vaccine schedule. Strange it may seem, the same logic appears to hold true for the enhanced efficacy of a single dose, a longer dose interval and one half and a full dose regimen.
Interestingly, the high efficacy of Russian sputnik ‘V’ vaccine (91 percent) also demonstrates the same logic. To explore this possibility, Oxford and Gameleya institute of Russia declared a joint clinical trial on a hybrid vaccine schedule in which one dose each of Oxford Astrazeneca and Russian Gamaleya vaccine. If successful, these combinations may become ground breaking discoveries in future vaccine research.
It should be noted here that Oxford vaccine uses modified Chimpanzee adenovirus as the vector to carry the Covidvirus spike protein components carrying genetic code inside recipients’ body, while Gamaleya vaccine uses inactivated human adenovirus for the same purpose. Hence these vaccines are categorized as viral-vector-vaccine where the inactivated virus vectors act as “viral postman” to deliver the Covid virus genetic code to the cells of the recipient subject.
The problem with a viral-vector vaccine is that while generating an immune response against the coronavirus’s spike protein, it will respond against the viral vector also. Such immune response produced by the first dose of vaccine may destroy or inhibit some of the “viral postman” of the booster second dose, before it can deliver the genetic code for reproduction, thus diminishing the effect in short interval.
Therefore a single dose is preferable for about 12 weeks because if the second dose is given earlier the viral postman will be affected adversely and the efficacy will suffer. If, however, the second dose is given after a longer interval of 12 or more weeks the mounted immune response does not affect the viral vector much and thus enhances efficacy. The central issue here for vaccine efficacy is the compromised, inhibited or destroyed ‘viral postman’s’ ability to deliver the corona virus protein to the cells after the second booster dose.
The same reasoning is applied to explain higher efficacy of reduced first dose and full second dose regimen. The first half dose may not allow for a strong anti-vector immune response to develop, thus helping the second dose to achieve its full booster potential and greater overall efficacy.
To escape from this problem of immune response destroying the carrier virus, Russian Sputnik V vaccine came up with a smart design of two different human Adenovirus vectors: Ad26 and Ad5. The immune response against one adenovirus is not so effective against the second hence its booster seems to work better, even though some adverse effect cannot be ruled out.
This is the reason for partnership of Oxford and Gamaleya for a hybrid vaccine. The likelihood of adversely affecting the viral vector should be even less between human and chimpanzee virus. The speculation is that the efficacy of hybrid vaccine will be higher than Russian vaccine and comparable to Pfizer (95 percent) or Moderna (94 percent), while doing away the limitations of very low temperature (-70, -20) cold chain storage of the later vaccines.
But these are conjectures for the future. Nepal has completed its first phase of Covishield vaccination program successfully and has paused for the second dose to be given after 28 days of the first one to this vaccinated population. The present challenge is to meticulously review the scientific evidence of past two weeks and make an informed decision by the government scientific body overseeing vaccination program.
I don’t see any reason why Nepal should not increase the dose interval to 12 weeks from the first dose. An alternative for more practical, cheaper and higher efficacious strategy for larger coverage has emerged just by readjusting the dose interval of Covishield vaccine which a resource constrained country like Nepal needs desperately.
You May Like This

Aug 21: 6 things to know by 6 PM
Your daily dose of missed important news of the day. ... Read More...

What Nepal needs is India's friendship and support for growth: Nepal PM Oli
In an exclusive interview to The Hindu, Mr. Oli says the bitterness of past relations have been put behind them,... Read More...

Helping women re-envision a better Nepal: Women LEAD Nepal
KATHMANDU,March 7: Women LEAD Nepal started as an idea that women and girls could change the trajectory of Nepal’s future and... Read More...







Just In
- NRB to provide collateral-free loans to foreign employment seekers
- NEB to publish Grade 12 results next week
- Body handover begins; Relatives remain dissatisfied with insurance, compensation amount
- NC defers its plan to join Koshi govt
- NRB to review microfinance loan interest rate
- 134 dead in floods and landslides since onset of monsoon this year
- Mahakali Irrigation Project sees only 22 percent physical progress in 18 years
- Singapore now holds world's most powerful passport; Nepal stays at 98th










Leave A Comment