
OR
The "Big Bang" of Alzheimer’s: Breakthrough study uncovers genesis of the disease
Published On: July 11, 2018 12:20 PM NPT By: Agencies
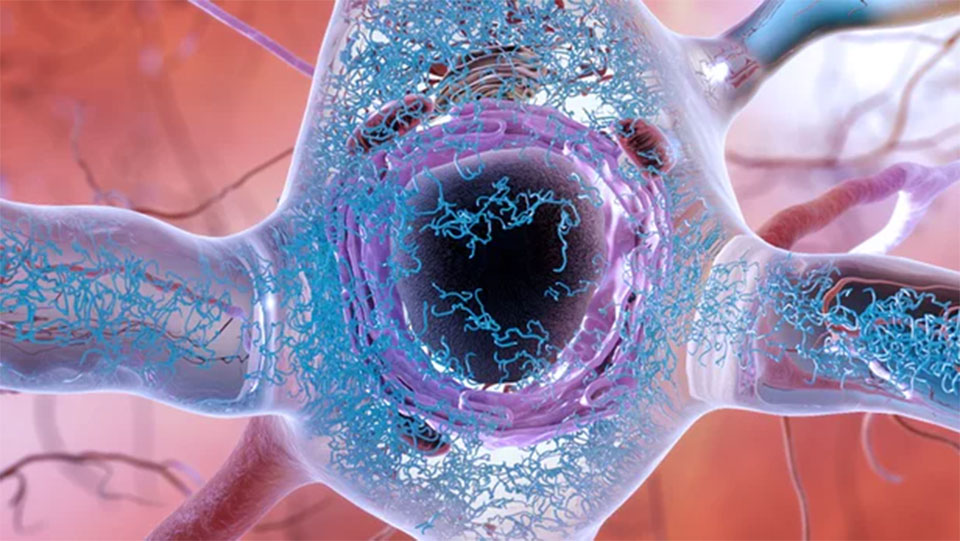
July 11: Impressive new research led by scientists at UT Southwestern, has discovered the earliest point in a neurodegenerative process that is thought to lead to dementia. The researchers describe the discovery as like finding the "Big Bang" of Alzheimer's disease, and it's hoped the work leads to new treatments and ways to detect the disease before major symptoms take hold.
"This is perhaps the biggest finding we have made to date," says Marc Diamond, a primary collaborator on this new study, "though it will likely be some time before any benefits materialize in the clinic. This changes much of how we think about the problem."
Much modern Alzheimer's research concentrates on a specific protein called amyloid beta, and the clumping of that protein is suspected as being the primary pathological cause of the disease's symptoms. But, after a long series of clinical trial failures in drugs designed to target those amyloid beta plaques, some scientists are turning their research attentions elsewhere.
This new research focuses on a different protein, called tau. These tau proteins have been found to form abnormal clumps in the brain, called neurofibrillary tangles, which can accumulate and kill neurons. Some researchers hypothesize that this is actually the primary causative source of Alzheimer's disease.
Until now it was not known how, or when, these tau proteins began to accumulate into tangles in the brain. It was previously believed that isolated tau proteins didn't have a distinctly harmful shape until they began to aggregate with other tau proteins. But the new research has revealed that a toxic tau protein actually presents itself as misfolded, exposing parts that are usually folded inside, before it begins to aggregate. It is these exposed parts of the protein that enable aggregation, forming the larger toxic tangles.
"We think of this as the 'Big Bang' of tau pathology," says Diamond. "This is a way of peering to the very beginning of the disease process. It moves us backward to a very discreet point where we see the appearance of the first molecular change that leads to neurodegeneration in Alzheimer's."
From here the research is set to take two different prospective pathways. One will first look at developing a simple diagnostic test to detect signs of this abnormal tau protein, either through a blood test, or less ideally a spinal fluid test. If these toxic tau proteins can be easily detected then clinicians may be able to diagnose Alzheimer's before major degenerative cognitive symptoms take hold.
The second research pathway flowing out of this major discovery involves investigating prospective drug treatments that could interrupt the tau aggregation process. The researchers point to a new drug called tafamidis, as an exciting example of a similar medicine designed to stabilize a protein that can clump and cause adverse symptoms.
Tafamidis was designed to delay impairment to nerve function caused by the toxic aggregation of a normally harmless protein called transthyretin and is currently approved for use in both Europe and Japan. However, the FDA has called for further clinical proof before approving the drug for use in the United States.
Now that this early alteration in the shape of tau molecules has been identified, researchers can more effectively focus on potential drug targets to inhibit the toxic aggregations at this stage.
"The hunt is on to build on this finding and make a treatment that blocks the neurodegeneration process where it begins," says Diamond. "If it works, the incidence of Alzheimer's disease could be substantially reduced. That would be amazing."
You May Like This

Not so big fish in a big pond
Krishna Prasad Sigdel’s book reads like a primer on Nepal’s foreign policy and international affairs. It also deals with the... Read More...

‘The Big Bang Theory’ renewed for two seasons
“The Big Bang Theory” just got a big renewal from CBS and Warner Bros. Television. ... Read More...

Infographics: Alzheimer’s disease
Infographics: Alzheimer’s disease ... Read More...




Just In
- Weather expected to be mainly fair in most parts of the country today
- 120 snow leopards found in Dolpa, survey result reveals
- India funds a school building construction in Darchula
- Exploring opportunities and Challenges of Increasing Online Transactions in Nepal
- Lack of investment-friendly laws raises concerns as Investment Summit approaches
- 550,000 people acquire work permits till April of current fiscal year
- Fixing a win by outlawing dissent damages democracy
- MoHP cautions docs working in govt hospitals not to work in private ones











_20220508065243.jpg)
Leave A Comment