
OR
Here is what experts, WHO and CDC have to say about J&J vaccine, safety and possible health problems
Published On: July 19, 2021 05:01 PM NPT By: Kunga Hyolmo
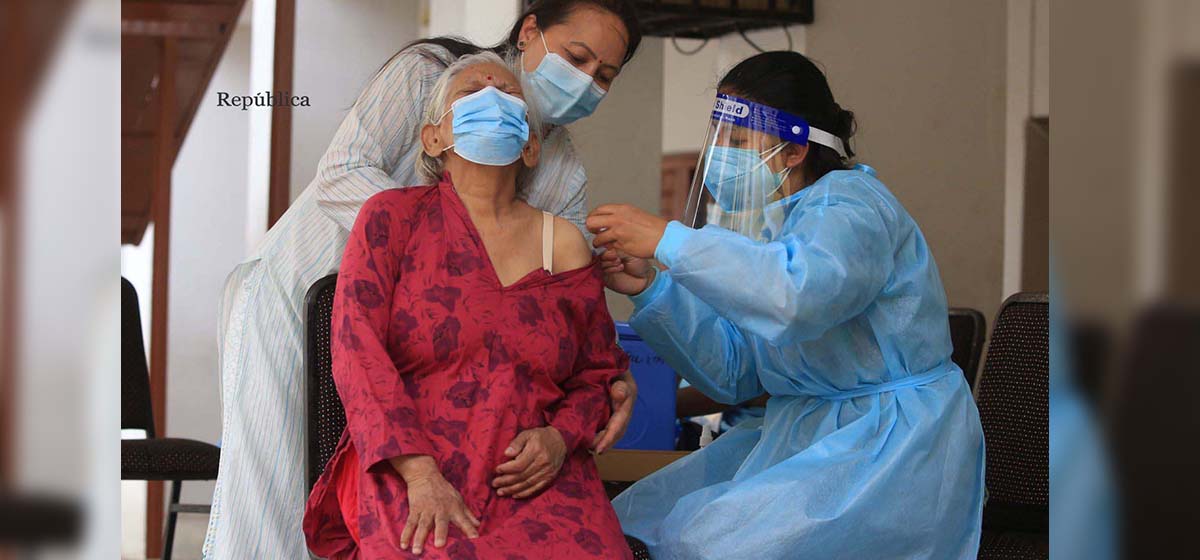
Vaccine is safe and effective: experts
KATHMANDU, July 19: With the government launching a fresh inoculation drive from Monday against COVID-19 using J&J vaccines received from the US, a section of people has raised questions about its efficacy and potential health risks. The questions and curiosities are natural, especially when new vaccines are being administered. Keeping this in mind, Republica has tried to address many of these questions and curiosities that ordinary people may have about this vaccine.
Nepal received 1.634 million doses of J & J vaccine from the US last month. The government is administering this vaccine to people aged between 50 to 55, who haven’t received any dose of any vaccine yet, differently-abled people (category ‘A’ and ‘B’), refugees currently living in Nepal and health workers and sanitation workers associated with health institutions. Here is what experts, WHO and Center for Disease Control and Prevention (CDC) in the US have to say about the J & J vaccine, its safety standards and possible health problems.
Who is the vaccine not recommended for?
The World Health organization (WHO) does not recommend individuals with a history of anaphylaxis to any component of the vaccine. Likewise, the vaccine is also not recommended to anyone suffering from high fever. Also, persons younger than 18 years of age should not receive the shot.
What does the CDC of the USA have to say about possible health problems?
Since use of COVID-19 vaccines began in the United States, scientists and doctors have constantly and carefully reviewed all reports of the vaccine's side effects and adverse events. As of April 23, 2021, more than 8 million doses of the J&J/Janssen COVID-19 vaccine had been administered in the United States. As of April 23, 2021, experts reviewing safety reports for this vaccine found 15 reports of women who got the J&J/Janssen COVID-19 Vaccine and later developed thrombosis with thrombocytopenia syndrome (TTS). The TTS is a serious condition that involves blood clots with low platelets.
These reports suggest an increased risk of the TTS one to two weeks after vaccination with the J&J/Janssen COVID-19 vaccine. As of April 23, 2021, the reports reviewed occurred in women between 18 and 59 years old, with a median of 37 years. These reports represent a reporting rate of 7 such events per 1 million vaccinations among women 18 through 49 years old and a rate of 0.9 per 1 million vaccinations among women 50 years and older. For all women, this is a rare adverse event. For women 50 years and older and men of all ages, the adverse event is even more rare. Reports show that symptoms of this adverse event started between 6 and 15 days after vaccination.
How effective is the vaccine?
According to the WHO, 28 days after inoculation Janssen Ad26.CoV2.S was found to have an efficacy of 85.4% against severe disease and 93.1% against hospitalization.
A single dose of Janssen Ad26.COV2.S was found in clinical trials to have an efficacy of 66.9% against symptomatic moderate and severe SARS-CoV-2 infection.
What are the side effects of the vaccines, if any?
According to Dr Sher Bahadur Pun, virologist at the Sukraraj Tropical and Infectious Disease Hospital at Teku, Kathmandu, rare cases of thrombosis with thrombocytopenia syndrome (TTS) and Guillain-Barre syndrome have been seen in some people receiving this vaccine in the US. Guillain-Barre syndrome is a rare disorder in which the human body's immune system attacks nerves. Weakness and tingling in extremities are usually the first symptoms. Likewise, TTS is an extremely rare condition characterized by abnormally low levels of platelets, also known as thrombocytes, in the blood. It is the most common coagulation disorder among intensive care patients and is seen in 20% of medical patients and a third of surgical patients. Similarly, body ache and high fevers are the minor side effects seen after receiving J&J Vaccine.
What are the myths regarding the vaccine?
1) The J&J vaccine is less effective at preventing hospitalization or death from COVID-19.
2) The J & J vaccine causes paralysis.
3) America donated it to Nepal because it had safety issues and was ineffective against COVID-19.
In comparison with other COVID-19 vaccines, where does the J & J vaccine stand?
Experts maintain that the COVID-19 vaccines cannot be compared head-to head due to the different approaches taken while developing it. But it is certain that all the COVID-19 vaccines that were given permission for use against the novel coronavirus disease are highly effective. These vaccines prevent severe conditions and hospitalization due to the disease.
Scientific studies conducted before the emergence of the Delta variant showed that the Sinovac and Sinopharm jabs were 50% and 79% effective, respectively, in preventing COVID-19 infections. Likewise, covishield vaccine has been found 70 percent effective against COVID-19 after receiving both the doses.
You May Like This
How long does protection from COVID-19 vaccines last?
NEW YORK, May 14: Experts don’t know yet because they’re still studying vaccinated people to see when protection might wear... Read More...
Biden gets COVID-19 vaccine, says ‘nothing to worry about’
NEWARK, Del, Dec 22: President-elect Joe Biden on Monday received his first dose of the coronavirus vaccine on live television... Read More...
Tuberculosis vaccine may be limiting COVID-19 deaths; dormitory screening urged
LONDON, July 12: The following is a brief roundup of some of the latest scientific studies on the novel coronavirus... Read More...




Just In
- Health ministry to conduct ‘search and vaccinate’ campaign on May 13
- Indian customs releases trucks carrying Nepali tea, halted across Kakarbhitta
- Silent period for by-election to begin from midnight
- SC issues short-term interim order to govt and TU not to take immediate action against TU legal advisor Khanal
- National consultation workshop advocates to scale up nutrition smart community in Nepal
- Patan High Court issues short-term interim order to halt selection process of NTB’s CEO
- NEPSE inches up 0.15 points; daily turnover increases to Rs 2.53 billion
- Bagmati Govt mandates tri-lingual signboards in offices













Leave A Comment